Global Journal of Medical Therapeutics
ISSN: e2687-4202
The Official Journal for Global Healthcare Activities Academy
Commentary
Year: 2019 I Volume: 1 I Issue: 1 I Pages: 5-7
Cabozantinib: cabometyx versus cometriq. Are they interchangeable?
Nagwa Ibrahim
Department of Pharmacy, Prince Sultan Military Medical City, Riyadh, Saudi Arabia
Correspondence Address:
Nagwa Ibrahim
Department of Pharmacy, Prince Sultan Military Medical City, Riyadh, Saudi Arabia
Email address: nag_ibrahim@hotmail.com
Date of online publication: 22 June 2019
Get permission for commercial use: contact us at editor@gjmt.net
https://doi.org/10.46982/gjmt.2019.101
Abstract:
Cabozantinib is an oral multi-tyrosine kinase inhibitor that targets vascular endothelial growth factor (VEGFR), mesenchymal epithelial transition factor (MET) and AXL. Cabozantinib is used for treating different types of cancer. It is available in the market in two different dosage forms with two different trade names. Cabometyx (cabozantinib) is available as 20mg, 40mg and 60mg film coated tablets and is approved by FDA for the management of renal cell carcinoma and hepatocellular carcinoma. However, cometriq (cabozantinib) is available as 20mg and 80mg capsules and is approved by FDA for treatment of thyroid cancer. Availability of two dosage forms for cabozantinib with different trade names for different indications is considered as potential for confusion and prescribing error. Implementation of good prescribing practice is essential in this case.
Successive governments have promoted the prescribing of drugs by generic rather than the trade name. Some drugs should not be prescribed generically for different reasons, for example when dosage forms of the products are not pharmaceutically equivalent. Cabozantinib is an example for this scenario. Cabometyx and cometriq are not bioequivalent.
In conclusion, each institution should have a generic prescribing policy. This policy must include drugs that cannot be prescribed generically. Based on posology, cabometyx and cometriq should not be used interchangeably but in case a patient must switch from cabozantinib capsules to cabozantinib tablets, the patient should continue with cabometyx using a dose not exceeding 60mg or to continue with the current cometriq dose whichever is lower. Do NOT substitute cabometyx tablets with cometriq capsules.
Key words: cabozanitinib, cabometyx, cometriq, pharmacokinetics, interchangeable.
How to cite this article:
Ibrahim N. Cabozantinib: cabometyx versus cometriq, Are they interchangeable? Glob J Med Therap 2019,1: 9-16.
1- Introduction:
Cabozantinib is a multi-tyrosine kinase inhibitor approved by FDA and available in the market in two dosage forms. Cabometyx (cabozantinib) is available as 20mg, 40mg and 60mg film coated tablets and cometriq (cabozantinib) is available as 20mg and 80mg capsules. Each dosage form is approved by FDA for a different indication.
Availability of cabozantinib in the market as two different trade products while being approved by FDA for different types of cancer is considered as potential source of confusion and prescribing error among healthcare professionals. Prescription errors could be minor and harmless or might result in life threatening situations.
This article is of significant importance for daily clinical practice as it clarifies the difference between cabometyx and cometriq, mainly for healthcare professionals dealing with oncology patients in order to avoid medication errors and ensure patient safety.
2- Generic prescribing practice:
Generic prescribing is promoted in many countries as an indicator of good prescribing practice. Using the generic name means prescribing the drug using its active ingredient rather than its brand name. There are many reasons to prescribe drugs using generic rather than brand names, such as difficulty to remember different brand names, no need to remember which brand is currently subsided, less potential for confusion among healthcare professionals and medication errors, in addition to the use of less expensive medication brands more often, pharmacists can dispense the medication in stock without a need for consultation and providing a guide to the drug’s pharmacology and chemical class, all which can be performed easier using the generic name [1-3].
It is not always possible to prescribe generically. Brand names should be used where it is clear that prescribing generically will lead to confusion for the dispending drug or create problems with bioavailability. This scenario applies in the following situations [1-6]:
1. The product is modified release: substitution of a modified release product with standard release formulations might not be advisable once treatment started as the composition and pharmacokinetics are difficult to standardize. This applies to some drugs as theophylline and isosorbide mononitrate.
2. The product has a narrow therapeutic range: in drugs with an efficacy and/or toxicity that are critically dependent on plasma concentration, the allowable differences in bioavailability between the reference and generic product in bioequivalence testing may result in changes in clinical effect between brands, although this is unlikely. Examples for such products are lithium carbonate and ciclosporin.
3. The delivery systems or dosage forms of the products are not pharmaceutically equivalent: e.g. cabometyx tablets and cometriq capsules, both products have the same active ingredient which is cabozantinib but they are not bioequivalent.
4. Combined preparations: e.g. hormone replacement therapy and oral contraceptive.
5. The same drug is used for different and separately branded indications: for example cabometyx is approved for RCC and HCC and cometriq is approved for MTC. Both brands have cabozantinib as active ingredient.
3- Cabozantinib FDA approved indications:
Table 1 - Cabozantinib approved indications
4- Cabozantinib dosing/ administration:
Table 2 - Cabozantinib dosing and administration
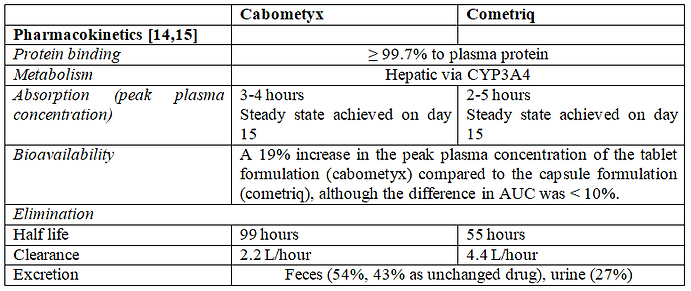
5- Cabozantinib pharmacokinetics:
Table 3 - Cabozantinib pharmacokinetics
Pharmacokinetics (PK) is a relevant and obvious biomarker that could be used to optimize treatment through therapeutic drug monitoring (TDM). The purpose of this discussion is to integrate the available PK data about cabozantinib tablets and capsules into practical recommendations for personalization of treatment.
A pharmacokinetics study of cabozantinib tablets and capsules formulations has been conducted by Nguyen and colleagues [8]. Results revealed that plasma exposure values were similar (<10% difference) for both formulations and the 90% confidence intervals (CIs) around the ratio of geometric least square means (GLSM) were within the accepted bioequivalence limits of 80-125%. However, the GLSM for Cmax was 19% higher for the tablet formulation and the upper 90% CI for the ratio of GLSM (131.65%) was beyond the 80-125% range. This finding indicates that tablets and capsules formulations failed to fulfill the bioequivalence study acceptance criteria and should not be used interchangeably [14].
Another pharmacokinetics analysis indicated similar steady-state exposures (Ctrough,ss) at different doses among patients with metastatic thyroid cancer (MTC) (140 mg capsule), renal cell carcinoma (RCC) (60 mg tablet). The apparent oral clearance estimated by patients’ pharmacokinetics model was 4.4 L/hour in MTC and 2.2 L/hour in RCC. These were unexpected results as Cmax and AUC of the tablet formulation (Cabometyx) and the capsule formulation (Cometriq) were similar following a single 140-mg dose [15, 16].
6- Cabometyx and cometriq, are they interchangeable:
Cabometyx (cabozantinib) tablets and cometriq (cabozantinib) capsules are not bioequivalent and should not be used interchangeably. If a patient must switch from cabozantinib capsules to cabozantinib tablets, the patient should continue at a cabometyx dose not to exceed 60mg or the current cometriq dose (whichever is lower). Do NOT substitute cabometyx tablets with cometriq capsules[17,18].
7- Conclusion and recommendations:
It is important to have postmarketing evaluation for the potential factors impacting pharmacokinetics of cabozantinib, such as patient population, formulations and doses in an integrated population pharmacokinetics model.
Each institution should have a generic prescribing policy. This policy must include drugs that cannot be prescribed generically. Cabozantinib should not be prescribed generically. It must be prescribed as cabometyx tablets or cometriq capsules as indicated.
Based on posology, cabometyx and cometriq should not be used interchangeably. In case a patient must switch from cabozantinib capsules to cabozantinib tablets, the patient should continue with cabometyxat a dose not exceeding 60mg or continue with the current cometriq dose, whichever is lower. Do NOT substitute cabometyx tablets with cometriq capsules.
Conflict of interest: The author discloses no conflict of interest.
Author contributions: Author agrees to be accountable for the content of the work.
Funding: The author disclosed no funding to this study.
Acknowledgments: None
References:
1- Walley T, Burrill P. Generic prescribing: time to regulate the market? BMJ. 2000;320 (7228): 131-132. doi: 10.1136/bmj.320.7228.131.
2- Rheinstein P. Therapeutic inequivalence. Drug Saf 1990;5(Suppl 1):114-9. https://doi.org/10.2165/00002018-199000051-00018.
3- Mott D, Cline R. Exploring generic drug use behaviour: The role of prescribers and pharmacists in the opportunity for generic drug use and generic substitution. Med Care 2002;40(8):662-74. doi:10.1097/01.MLR.0000020926.85284.8E.
4- Josemir W Sander, Philippe Ryvlin, Hermann Stefan, Daniel R Booth & Jürgen Bauer. Generic substitution of antiepileptic drugs. Expert Review of Neurotherapeutics. 2010; 10:12, 1887-1898. doi: 10.1586/ern.10.163.
5- Andrade C. Bioequivalence of generic drugs. J Clin Psychiatry. 2015; 76(9):e1130-1. doi: 10.4088/JCP.15f10300.
6- Sarpatwari A, Choudhry NK, Avorn J, Kesselheim AS. Paying Physicians to Prescribe Generic Drugs and Follow-On Biologics in the United States. PLoS Med. 2015; 12(3): e1001802. https://doi.org/10.1371/journal.pmed.1001802.
7- Yakes FM, Chen J, Tan J, Yamaguchi K, Shi Y, Yu P, Qian F, Chu F, Bentzien F, Cancilla B, Orf J, You A, Laird AD, Engst S, Lee L, Lesch J, Chou YC, Joly AH Mol. Cabozantinib (XL184), a novel MET and VEGFR2 inhibitor, simultaneously suppresses metastasis, angiogenesis, and tumor growth. Mol Cancer Ther. 2011; 10(12):2298-308. doi: 10.1158/1535-7163.MCT-11-0264.
8- Cella D, Escudier B, Tannir NM, Powles T, Donskov F, Peltola K, Schmidinger M, Heng DYC, Mainwaring PN, Hammers HJ, Lee JL, Roth BJ, Marteau F, Williams P, Baer J, Mangeshkar M, Scheffold C, Hutson TE, Pal S, Motzer RJ, Choueiri TK. Quality of Life Outcomes for Cabozantinib Versus Everolimus in Patients With Metastatic Renal Cell Carcinoma: METEOR Phase III Randomized Trial. J Clin Oncol. 2018; 36(8):757-764. doi: 10.1200/JCO.2017.75.2170.
9- Choueiri TK, Halabi S, Sanford BL, Hahn O, Michaelson MD, Walsh MK, Feldman DR, Olencki T, Picus J, Small EJ, Dakhil S, George DJ, Morris MJ. Cabozantinib Versus Sunitinib As Initial Targeted Therapy for Patients With Metastatic Renal Cell Carcinoma of Poor or Intermediate Risk: The Alliance A031203 CABOSUN Trial. J Clin Oncol. 2017; 35(6):591-597. doi: 10.1200/JCO.2017.75.2170.
10- Powles T, Albiges L, Staehler M, Bensalah K, Dabestani S, Giles RH, Hofmann F, Hora M, Kuczyk MA, Lam TB, Marconi L, Merseburger AS, Fernández-Pello S, Tahbaz R, Volpe A, Ljungberg B, Bex A. Updated European Association of Urology Guidelines Recommendations for the Treatment of First-line Metastatic Clear Cell Renal Cancer. Eur Urol. 2017 Dec 7. pii: S0302-2838(17)31001-1. doi: 10.1016/j.eururo.2017.11.016.
11- Abou-Alfa GK, Meyer T, Cheng AL, et al. Cabozantinib in patients with advanced and progressing hepatocellular carcinoma. N Engl J Med. 2018; 379 (1):54-63. doi: 10.1056/NEJMoa1717002.
12- Schlumberger M, Elisei R, Müller S, et al. Overall survival analysis of EXAM, a phase III trial of cabozantinib in patients with radiographically progressive medullary thyroid carcinoma. Ann Oncol. 2017; 28(11): 2813-2819. doi: 10.1093/annonc/mdx479.
13- Cochin V, Gross-Goupil M, Ravaud A, Godbert Y, Le Moulec S. Cabozantinib: Mechanism of action, efficacy and indications. Bull Cancer. 2017; 104(5): 393-401. doi:10.1016/j.bulcan.2017.03.013.
14- Nguyen L, Benrimoh N, Xie Y, Offman E, Lacy S. Pharmacokinetics of cabozantinib tablet and capsule formulations in healthy adults. Anticancer Drugs. 2016; 27(7):669-78. doi: 10.1097/CAD.0000000000000366.
15- Food and Drug Administration. Center for Drug Evaluation and Research Cabozantinib Clinical Pharmacology and Biopharmaceutics Review. http://www.accessdata.fda.gov/drugsatfda_docs/nda/2012/203756Orig1s000ClinPharmR.pdf . Accessed 14-6-2019.
16- Singh H, Brave M, Beaver JA, Cheng J, Tang S, Zahalka E, Palmby TR, Venugopal R, Song P, Liu Q, Liu C, Yu J, Chen XH, Wang X, Wang Y, Kluetz PG, Daniels SR, Papadopoulos EJ, Sridhara R, McKee AE, Ibrahim A, Kim G, Pazdur R. U.S. Food and Drug Administration Approval: cabozantinib for the treatment of advanced renal cell carcinoma. Clin. Cancer Res. 2017; 23, 330–335. doi: 10.1158/1078-0432.CCR-16-1073.
17- Cabozantinib summary of product characteristics. Ema.Europe.eu website. https://www.ema.europa.eu/en/documents/product-information/cabometyx-epar-product-information_en.pdf Accessed June 14, 2019.
18- Emc – cabometyx website https://www.medicines.org.uk/emc/product/4407/smpc Accessed June 14, 2019.